If you have ever sat across from a provider and been told your fertility workup looks “normal” while you are still not pregnant, you are not imagining the gap. The standard preconception evaluation in most OB/GYN offices was designed to confirm a few binary questions: Are you ovulating? Is your thyroid in range? Is there a structural issue?
Those questions matter. They are also incomplete.
A truly complete hormonal workup answers a different, harder question: What is the full endocrine and metabolic environment in which you are trying to conceive a baby? That requires looking at hormones most workups never order, drawn on cycle days that most workups never specify, and interpreted through frameworks that most workups never apply.
This is the evaluation we do at Materna. It is grounded in current reproductive endocrinology guidelines from the American Society for Reproductive Medicine, layered with the functional medicine lens that catches what conventional bloodwork tends to miss.
Below is what that workup looks like, step by step.
Step one: the core ovarian reserve panel
The first three hormones must be drawn together, in the early follicular phase, on cycle days two through four. This timing is not optional. It is the only window in which these values are interpretable.
Follicle stimulating hormone (FSH) is released by the pituitary to recruit ovarian follicles each cycle. As ovarian reserve declines, the ovary produces less feedback (less inhibin, less estradiol), so the pituitary has to push harder. An FSH below 10 IU/L suggests adequate reserve. Higher values suggest diminished ovarian reserve.
Estradiol (E2) must be drawn on the same day as FSH, because the two values interpret each other. An early follicular estradiol below 80 pg/mL is reassuring. If estradiol is already elevated on cycle day three, it can suppress FSH into a falsely reassuring range, masking declining reserve. This is why checking FSH alone is misleading. Both together tell the truth.
Anti Müllerian hormone (AMH) is produced by the granulosa cells of small antral follicles and is the most direct serum measure of the primordial follicle pool. It can be drawn on any cycle day, which makes it operationally simple. Above 1 ng/mL suggests adequate quantity. Above 6 ng/mL suggests PCOS and predicts a strong stimulation response. AMH measures quantity of eggs, not quality. Age remains the strongest predictor of egg quality and live birth, and AMH can be artificially low in women on hormonal contraceptives or with elevated BMI.

Step two: confirming ovulation
Progesterone is the test most women have heard of, and the one most workups stop at. It is drawn approximately seven days before expected menses, around cycle day 21 in a 28 day cycle. A value of 3 ng/mL or higher confirms ovulation occurred.
This is genuinely useful information. It is also the floor of a workup, not the ceiling. Confirming you ovulated tells us nothing about why a cycle is irregular, why implantation is failing, or why hormones are dysregulated upstream.
Step three: when ovulation is irregular, look upstream
If a patient has irregular cycles, missed periods, or confirmed anovulation, the next layer of testing identifies the cause.
Thyroid stimulating hormone (TSH) screens for thyroid dysfunction, which accounts for two to three percent of anovulatory infertility. We test this routinely, and we test the full thyroid panel when the clinical picture warrants it.
Prolactin is drawn when there is galactorrhea (milky nipple discharge), oligomenorrhea, or amenorrhea. Hyperprolactinemia, often from a small benign pituitary adenoma, suppresses GnRH pulsatility and shuts down ovulation. It accounts for roughly 13 percent of anovulatory cases.
Luteinizing hormone (LH) is interpreted in context. An LH to FSH ratio above 2 to 1 supports a PCOS diagnosis. Very low LH alongside low FSH suggests hypothalamic suppression. Very high LH alongside high FSH suggests primary ovarian insufficiency.
Total and free testosterone, plus DHEA sulfate (DHEA-S) are drawn when PCOS or androgen excess is suspected. Elevated androgens drive anovulation, hirsutism, and acne. DHEA-S specifically tells us whether the androgen excess is coming from the adrenal glands or the ovaries, which changes the treatment approach entirely.
17 hydroxyprogesterone (17 OHP) is the screening test for nonclassic congenital adrenal hyperplasia, a condition that mimics PCOS and accounts for up to 10 percent of women with hyperandrogenism. It must be drawn in the morning, in the follicular phase, because levels rise in the luteal phase and across the day. A basal value above 200 ng/dL warrants follow up testing with an ACTH stimulation test.
Step four: fasting insulin, the hormone almost no one orders
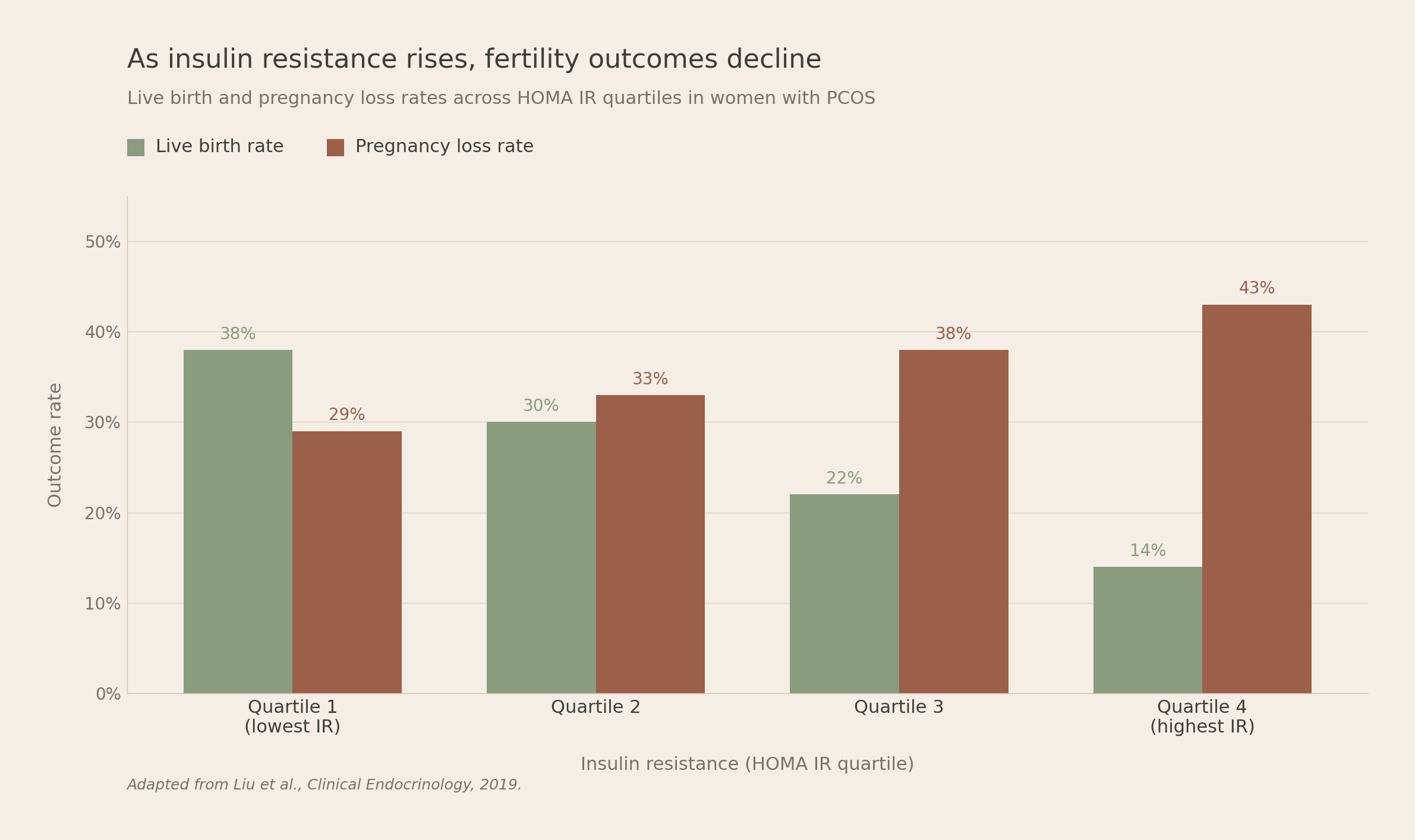
This is where conventional fertility workups consistently fall short.
Fasting insulin is not part of the traditional ASRM panel. It should be. Insulin resistance is one of the most common, most modifiable, and most under recognized drivers of subfertility, and it is invisible to a normal fasting glucose.
In PCOS, hyperinsulinemia plays a central pathogenic role. Elevated insulin stimulates ovarian theca cells to produce excess androgens, prevents the dominant follicle from being selected each cycle (creating anovulatory states), recruits too many small follicles (creating polycystic morphology), and reduces oocyte quality, embryo quality, and endometrial receptivity.
A 2022 Mendelian randomization study found that genetically determined higher fasting insulin was causally associated with greater odds of infertility in women, with an odds ratio of 1.60. In women with PCOS undergoing ovulation induction, rising insulin resistance correlates with lower ovulation, lower conception, lower live birth, and higher pregnancy loss in a clear dose dependent pattern.
Critically, insulin resistance occurs in both lean and obese women with PCOS. We have seen patient after patient with a normal BMI, a normal fasting glucose, and a fasting insulin that tells a completely different story. Identifying these patients early opens up evidence based interventions: targeted nutrition, inositol, sometimes metformin, that improve ovulation rates and reduce androgen levels before more invasive treatment is considered.
Step five: cortisol, sleep, and the HPA axis
Cortisol is not a routine fertility test, and a single random cortisol level is not clinically useful. But the hypothalamic pituitary adrenal axis profoundly influences reproduction, and chronic dysregulation matters.
When cortisol is chronically elevated, whether from prolonged stress, HPA dysfunction, or true Cushing syndrome, the reproductive axis is suppressed at multiple levels. CRH and ACTH derived peptides inhibit GnRH. Glucocorticoids suppress LH release from the pituitary. Cortisol directly inhibits ovarian estrogen and progesterone production. And glucocorticoids damage endometrial stem cell function, which impairs implantation.
Studies show that elevated cortisol across all phases of the menstrual cycle correlates with reduced estradiol secretion, an attenuated preovulatory LH surge, and lower conception rates.
Sleep is the most powerful and most overlooked modulator of cortisol. A single night of fragmented sleep elevates evening cortisol and flattens the normal diurnal decline. Chronic sleep deprivation, shift work, and circadian disruption are associated with menstrual irregularity, anovulation, early pregnancy loss, and reduced ART success.
This is why we ask about sleep at every preconception visit, and why we use four point salivary or urinary cortisol mapping (the DUTCH test) when the clinical picture suggests HPA dysregulation. Counseling on sleep hygiene, seven to nine hours, consistent timing, minimizing shift work where possible, is one of the most evidence based and underutilized fertility interventions in medicine.
What we do not test, and why
Patients sometimes arrive with lab orders from social media or a wellness influencer that include hormones we explicitly do not draw as part of a standard fertility workup. To name a few:
Pregnenolone is a precursor steroid sitting upstream of progesterone, cortisol, and DHEA. It does not correlate with specific ovulatory or reserve defects, and no major guideline recommends its measurement. It is too far upstream to be actionable.
Inhibin B was once proposed as an ovarian reserve marker. ASRM now explicitly states it is not a helpful tool to assess ovarian reserve and does not recommend it.
The clomiphene citrate challenge test is obsolete, replaced by AMH and antral follicle count.
Random cortisol, as discussed above, is not interpretable in isolation. Cortisol follows a strong diurnal rhythm and has to be measured at multiple time points or via specific protocols.
A complete workup is not a maximalist workup. It is the right workup, with each test ordered for a specific clinical reason.
The complete panel, summarized

What this looks like at Materna
At Materna, every patient trying to conceive receives this complete workup, drawn on the right cycle days, interpreted through both a traditional reproductive endocrinology framework and a functional medicine framework simultaneously. We do not stop at “your day 21 progesterone is normal.” We ask what your insulin is doing, what your cortisol is doing, what your full thyroid picture looks like, and how your adrenal androgens compare to your ovarian androgens.
Because here is what we have learned: the women who come to us after months or years of being told everything looks fine, are almost never women whose hormones have nothing to say. They are women whose hormones were never fully asked.
Ready to take the next step?
Our preconception guide walks you through the full evaluation framework we use at Materna, including the questions to ask your provider before your next bloodwork, what each value means, and how to interpret your results in context.
Download The Materna Preconception Guide
Evidence informed. Provider written. Built for women who want answers, not platitudes.
We're here for you - from the start
We’re redefining maternal health by replacing the ER with a welcoming, evidence-based care environment that centers you: your body, your symptoms, your experience.
Schedule a visit
Compassionate support for every stage of pregnancy
Contact
Connect with our dedicated support team
Phone
Direct line for immediate healthcare guidance
Office
Women's health center in
130 7th Ave S NY, NY 10014
130 7th Ave S NY, NY 10014
Related blogs and articles

Inside Materna Health's Grand Opening: Launching same day OB/GYN care
Materna Health celebrated its grand opening on April 23, 2026 in NYC's West Village. A look inside the night, the community, and the care we are building for early pregnancy and the forgotten in between.

Iron in Pregnancy: Why It Matters and When an IV Iron Infusion Is the Right Choice
Iron deficiency affects up to 70 percent of pregnancies and is routinely missed. Learn why iron matters, why oral iron is often not enough, and how an IV iron infusion at Materna can help you feel like yourself again.

Why Your Fertility Workup Is Probably Incomplete
Why Your Fertility Workup Is Probably Incomplete | Materna Health

